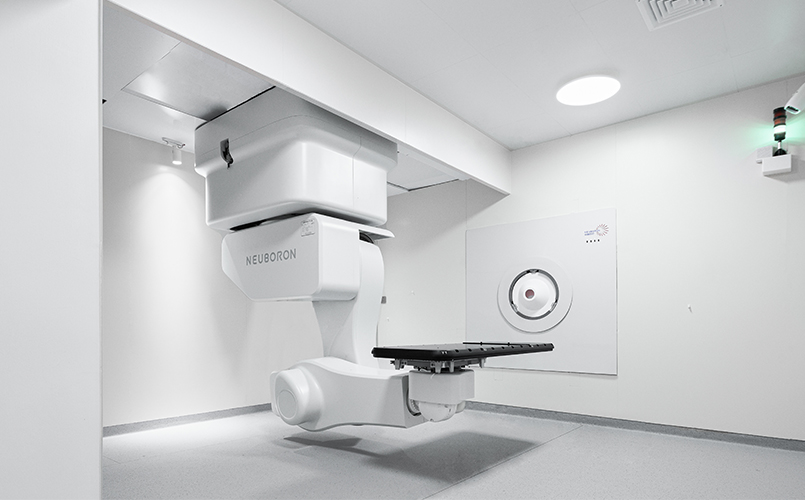
The Xiamen Humanity Hospital BNCT Center (XHH BNCT Center), the first BNCT clinical center and research in China has successfully completed the construction and delivered the stable epithermal neutron beam at 4 mA and 2.3 MeV, equaling to about 27% neutron yield at full power (2.5 MeV and 10 mA). Now formal animal pre-clinical study using mice, rat, and beagle is under way. This most advanced accelerator-based BNCT facility designed and developed by Neuboron Medical Group.
The XHH BNCT Center is a two-story high, and two-story underground building, which is dedicated to BNCT clinical and research purposes. It is fully designed to safely operate two horizontal beams and one vertical beam using the NeuPexTM Block-I AB-BNCT system, within a very compact space. In addition to the 3 treatment rooms, it has correspondingly 3 simulation rooms, in order to facilitate the patient positioning work and increase the patient turnover rate. It is expected to offer more than 4000 irradiations per year, which equals to more than 2000 patients. XHH BNCT Center is also well equipped with radiobiology, chemistry, ICP-MS, medical physics laboratories, and HPCs, providing ideal environment for scientific research. XHH BNCT Center will open for external research, and provide research funds to young researchers who have new ideas in BNCT.
XHH BNCT Center started its construction in May 2019, and completed the construction in late 2020 amid the global COVID-19 pandemic. Meanwhile, Neuboron's AB-BNCT system NeuPexTM began installation in September 2020, the 1st horizontal beam has been successfully installed, and the vertical beam will be installed in 2023. XHH BNCT Center has successfully tested its first epithermal neutron beam on the 5th August 2021, at 4 mA and 2.3 MeV, equaling to about 27% neutron yield at full power (2.5 MeV and 10 mA). The preliminary measurement shows the epithermal neutron flux is greater than 3×108 n/cm2/s at the beam exit, and greater than 7×108 n/cm2/s at the mice irradiation position. Then the first animal irradiation has been successfully performed on the 28th August 2021 using mice with and without tumor bearing, under GLP-like environment. The group of 500 mg/kg BPA infused mice showed superior tumor control performance. Currently, the beam is under commissioning and V&V for higher beam current and energy, preparing for the clinical trial.
About BNCT
BNCT is a combination treatment based on the reaction that occurs when a non-toxic compound containing boron-10 is irradiated with a low-energy neutron beam. BNCT differs radically from other radiation therapy and shows promise in becoming the next-generation cancer treatment. Research has shown BNCT has the capability of killing cancer cells that are resistant to traditional radiation therapy with limited harm to healthy tissue. Current advances in both neutron radiation technology and medicinal boron drug targeting are enabling BNCT’s potential to improve patient care while also improving treatment economics. To date, approximately 2,000 patients have been treated with BNCT at research sites worldwide.
About Neuboron Medical Group
Neuboron Medical Group was founded in 2014 focusing on the development of BNCT total solution for hospital users, including a complete set of accelerator-based BNCT system, TPS, theranostic boron or gadolinium carriers (e.g., BPA and F-BPA), as well as facility design and construction. Currently, it has developed its own BPA and F-BPA in China. The F-BPA is currently undergoing clinical study in the famous Peking Union Medical College Hospital using nucleophilic radiosynthesis.